GHS Classification: Pyr. Liq. 1 (GHS02), Flam. Liq. 1 (GHS02), Acute Tox. 2 (Inhalation) (GHS06), Skin Corr. 1A (GHS05), Eye Dam. 1 (GHS05), STOT SE 3 (GHS08), Aquatic Acute 1 (GHS09), Aquatic Chronic 1 (GHS09)
Hazard Statements: Catches fire spontaneously if exposed to air; Extremely flammable liquid and vapour; Fatal if inhaled; Causes severe skin burns and eye damage; May cause respiratory irritation; Very toxic to aquatic life with long lasting effects
UN Number: UN 3254
Hazard Class: 4.2 (Spontaneously combustible); Subsidiary hazard 6.1 (Toxic inhalation hazard)
Packing Group: I

Tributylphosphine CAS 998-40-3
Aure Chemical Delivers Excellence in Tributylphosphine
Tributylphosphine (TBP) (CAS 998-40-3) is a clear, colorless to pale yellow liquid that is one of the most widely utilized tertiary alkylphosphines in synthetic chemistry. It is characterized by its strong, pungent, and highly characteristic garlic-like odor. TBP is valued for its powerful reducing capabilities and its function as a soft, sterically non-hindered ligand for transition metals, particularly palladium and nickel. Its solubility profile and relatively low cost make it a practical choice for large-scale industrial and laboratory synthesis. TBP is a crucial reagent in many carbon-carbon and carbon-heteroatom bond-forming reactions.
Basic Information of TBP
Meticulously produced and rigorously tested to meet stringent quality standards. We ensure exceptional purity and consistent performance, essential for your critical applications:
| CAS No.: | 998-40-3 |
|---|---|
| EC No.: | 213-651-2 |
| Linear Formula: | C12H27P |
| Molecular Weight: | 202.32 |
| Appearance: | Colorless to pale yellow, clear liquid |
| Odor: | Strong, highly pungent, garlic-like |
| Melting Point: | -65 °C |
| Boiling Point: | 150 °C50 mm Hg(lit.) |
| Density: | 0.81 g/mL at 25 °C(lit.) |
| Solubility: | Insoluble in water; highly soluble in organic solvents (toluene, hexane, THF). |
| Flash Point: | 99 °F |
| RIDADR: | UN 3254 4.2/PG 1 |
| Chemical Structure: | 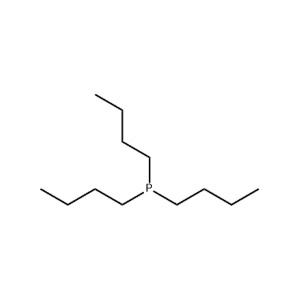 |
Application Overview
Tributylphosphine's versatility stems from its strong nucleophilicity, high ligand properties, and reducing power, making it key in various high-demand chemical syntheses:
Catalysis (Transition Metal Ligand): It serves as a fundamental phosphine ligand in many industrial transition-metal catalyzed coupling reactions, including various Suzuki, Heck, and Negishi couplings, particularly in cross-coupling reactions where moderate steric bulk is preferred.
Reducing Agent: Used as a non-metallic reducing agent in reactions such as the Staudinger reaction (reduction of azides to amines) and the Mitsunobu reaction (in combination with diethyl azodicarboxylate).
Polymerization: TBP is employed as a specialized catalyst or initiator in certain polymerization processes, particularly for ring-opening and addition polymerizations, influencing the molecular weight and structure of the final polymer.
Pharmaceutical Intermediates: It is a crucial reagent in the synthesis of complex pharmaceutical and agrochemical intermediates, often for forming specific functional groups or constructing complex molecular skeletons.
Why Choose Aure Chemical for Tributylphosphine?
Aure Chemical offers Tributylphosphine (CAS 998-40-3) with specialized quality assurance protocols centered on maximizing its reactivity, purity, and safe handling for synthetic chemistry.
Minimizing Phosphine Oxide Content: The primary degradation product, Tributylphosphine Oxide (TBPO), severely inhibits TBP's catalytic activity. We guarantee TBPO content below 0.1%, ensuring maximum ligand efficiency and reducing reagent consumption in sensitive catalytic cycles.
Inert Gas and Specialized Packaging: Due to TBP's extreme sensitivity to oxygen (leading to immediate oxidation), we utilize high-integrity, seamless stainless steel containers or glass ampoules under a rigorously maintained argon atmosphere. This packaging guarantees the integrity and purity of the reactive phosphine upon delivery.
Batch-Specific Spectroscopic Analysis: Every batch is accompanied by detailed ${}^{31}\text{P NMR}$ data alongside the Certificate of Analysis (CoA). This provides a direct, highly accurate measure of phosphine purity, giving chemists absolute confidence in the material's identity and reactivity.
Controlled Supply Chain for Safety: We adhere to the strictest global regulatory standards for handling and shipping corrosive and flammable materials (UN 2920). Our specialized logistics team ensures safe, compliant, and timely delivery directly to your secure storage facilities.
Hazards Classification





Packaging, Logistics & Compliance Capabilities
Aure Chemical provides flexible packaging solutions, compliant logistics arrangements, and controlled storage practices to support laboratory-scale, pilot-scale, and commercial-scale global supply.
Packaging Options
- 25 kg / 50 kg bags (fiber drum, aluminum bag, HDPE drum)
- 100 kg / 200 kg steel or HDPE drums
- 500 kg / 1000 kg IBC containers
- Bulk liquids in ISO tank containers
- Customized packaging upon request
Transportation Modes
- Sea freight (FCL / LCL)
- Air freight for selected products
- Road and rail transportation
- Dangerous & non-dangerous cargo handling
- Incoterms: EXW, FOB, CFR, CIF, DAP, DDP
Storage & Handling
- Dry, cool, and well-ventilated warehouses
- Segregation by hazard class
- FIFO inventory management
- Temperature-controlled storage if required
- Regular inspection & labeling control
Documentation & Compliance
- Safety Data Sheet (SDS)
- Certificate of Analysis (COA)
- Dangerous Goods Declaration (DGD)
- Packing List & Commercial Invoice
- IMDG / IATA / ADR compliant labeling
For packaging customization or logistics consultation, please contact us.








